Estimated reading time: 5 minutes
Few plant diseases have shaped agricultural history quite like late blight of potato. Caused by the oomycete pathogen Phytophthora infestans, this destructive pathogen is best known for triggering the Irish potato famine in the 1840s. Yet it remains a very real threat to potato producers today.
Late blight not only slashes yields, but also forces growers to spend heavily on fungicides and other preventative measures just to keep crops alive.
A new lineage emerges
Although the disease had already been reported in South Africa in the late 1800s, no major outbreaks were recorded for many years. Before the mid-1990s, late blight was uncommon in most potato-growing regions, particularly in the warmer inland areas away from the coast.
The disease appeared only sporadically, and long sunny periods after wet weather often helped producers keep it under control with chemicals. This natural break in the disease cycle made late blight easier and cheaper to manage.
That sense of security disappeared almost overnight. In 1995 and 1996, severe late blight outbreaks swept through almost all of South Africa’s potato-producing regions and even affected some tomato crops. These epidemics raised alarm bells among researchers and prompted a national survey of the disease.
Twenty years have passed since that outbreak, and even though only sporadic, localised outbreaks have occurred in South Africa in recent years, a new survey was conducted to determine the status of the pathogen in the country. Earlier work has already shown worrying signs: More than 80% of the isolates tested were tolerant to metalaxyl, a fungicide widely used at the time.
These isolates all belonged to the P. infestans lineage US-1.
Isolates of the late blight pathogen are classified into different lineages using specific genetic markers. The 1996 to 2007 outbreaks in South Africa were caused by P. infestans clonal lineage US-1 (McLeod et al., 2001; Pule et al., 2013). However, a recent study has revealed that this lineage has been replaced by lineage EU23 (McLeod et al., 2024), indicating introduction of new genotypes in the country.
Tracking the pathogen
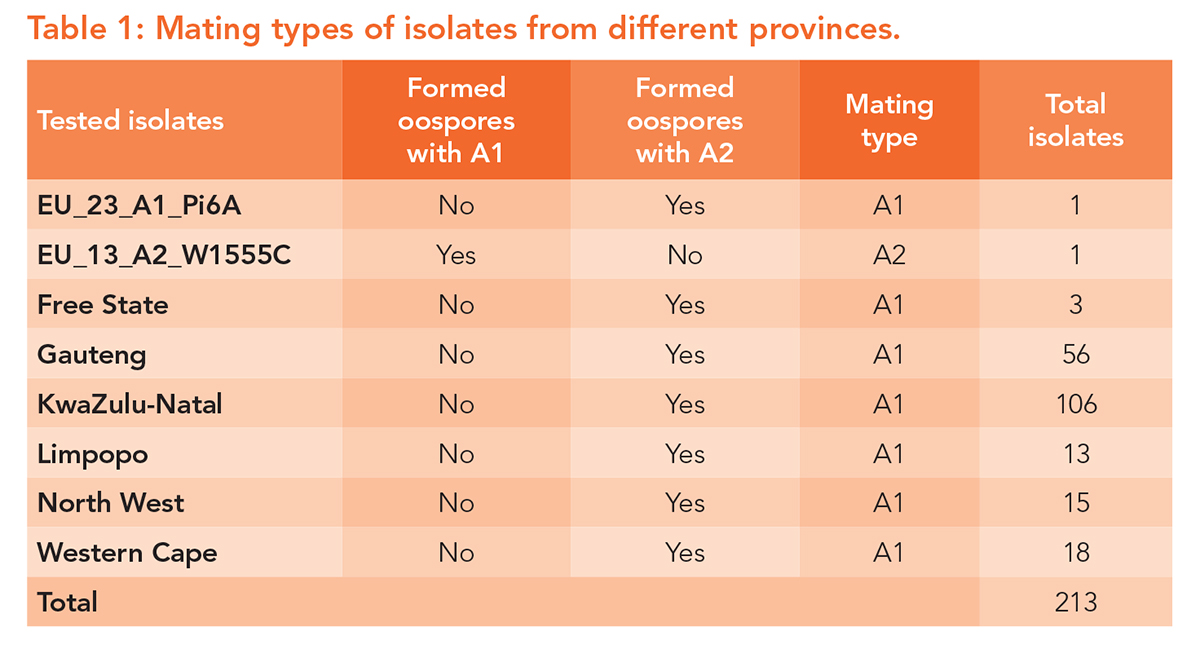
A new survey was therefore launched to understand what is happening within South Africa’s P. infestans populations. The aims were to determine which mating types are present, how aggressive the pathogen has become, and whether it is still sensitive to the fungicides producers rely on. There were no major nationwide outbreaks and despite repeated requests for samples, only 211 isolates from six provinces were obtained. Even so, these samples provided valuable insights.
To isolate the pathogen in the lab, leaves were incubated between tuber slices until sporulation occurred, after which sporangia were carefully transferred onto semi-selective rye agar, which proved to be the most effective method. While both pea agar and rye agar supported growth, rye agar proved essential for studying mating types because it reliably produced oospores, the structures needed to identify mating types.
All the isolates collected belonged to the A1 mating type (Table 1). This may sound like good news – and it is, for the moment. When both A1 and A2 mating types occur together, P. infestans can reproduce sexually, generating new genetic combinations. That kind of diversity allows the pathogen to evolve faster, potentially becoming more aggressive and more resistant to fungicides.
The absence of A2 suggests that South Africa’s late blight population is still relatively stable. Continued monitoring is crucial because the introduction of A2 could quickly change the disease landscape.
Tests and results
To assess how aggressive the pathogen is, six isolates from different regions were tested in both glasshouse trials and detached leaf experiments. All caused late blight symptoms (Figure 1), but not equally; some isolates were clearly more virulent than others.

Encouragingly, both testing methods produced almost identical results, confirming that the quicker, less resource-intensive detached leaf assay is a reliable tool for future screening.
Perhaps the most reassuring finding came from fungicide tests. Several products commonly used in South Africa, including azoxystrobin, amisulbrom, cymoxanil, fluazinam, and chlorothalonil, remain effective against P. infestans.
There is currently no evidence of widespread resistance to these chemicals.
Phosphoric acid products showed higher EC50 values, meaning higher doses were needed to suppress the pathogen in laboratory tests. However, this is not a major concern. These fungicides do not rely solely on directly killing the pathogen; they also strengthen the plant’s own defences. Although metalaxyl was not tested as part of the current study, McLeod et al. (2024) showed that the EU23 lineage is sensitive to this fungicide.
Staying one step ahead
Late blight may not be making headlines in South Africa right now, but it hasn’t gone away. This study shows that while the current situation is manageable, the pathogen still has the potential to adapt and spread.
Regular monitoring, careful fungicide resistance management, and early detection remain essential tools in protecting one of the country’s most important food crops. For potato producers, vigilance instead of complacency will be the key to staying ahead of this age-old enemy. – Elsie Cruywagen, Juanita Engelbrecht, Reneilwe Mohale and Chris Spies, Agricultural Research Council
For more information or references, send an email to CruywagenEM@arc.agric.za.
