Estimated reading time: 8 minutes
When potato certification started in South Africa in the 1990s, one of the strategic decisions of the potato industry to ensure the cost-effectiveness of certifying seed potatoes, was the establishment of Potato Laboratory Services (PLS). PLS consists of five regional laboratories currently situated in Piketberg, Douglas, Christiana, Pietermaritzburg and Pretoria, to facilitate testing of samples destined for certification.
The core function of PLS is the testing of official, statistical field samples from registered units, for the presence of Ralstonia spp. (causal of bacterial wilt of potato), as well as potato virus Y (PVY) and potato leafroll virus (PLRV).
The inclusion of additional functions at Plantovita in Pretoria (previously the Coen Bezuidenhout Seed Testing Centre or CBS) that could not be replicated cost-effectively at the other regional laboratories, made the certification process even more affordable without compromising the requirements set by the South African Seed Potato Certification Scheme (the Scheme).
To best serve the potato industry, standardising conduct and test methods within PLS is crucial. This is achieved inter alia through rigorous testing, adherence to approved methods, regular assessment
and auditing, identifying and managing risks to impartiality and undue pressures, and applying a
strict code of conduct. Monitoring standardisation is the responsibility of Plantovita as the controlling laboratory in PLS. Scheme requirements and the approved code of conduct restrict laboratories from
testing potato samples intended for the certification process.
Methods and procedures employed for certification purposes are based on European and Mediterranean Plant Protection Organization principles and current International Standards for Phytosanitary Measures. The accuracy of the outcome of any test method is determined not only by the properties of the test and the pathogen, but also by the representation of sampling.
Regulatory requirements
Because PLS fulfils regulatory functions in the potato industry and are service providers in agriculture, all laboratories are required to be registered at the Department of Agriculture, Land Reform and Rural Development (DALRRD) as laboratories testing for plants. In compliance with Regulations R110, if a regulated pathogen in a sample has been confirmed by testing, or is suspected to occur in a unit, DALRRD must be informed.
Laboratories registered with DALRRD may apply to the Independent Council for the Certification of
Seed Potatoes (ICCSP) for approval to function as testing laboratories under PLS, generating results that may be used in the certification process of seed potatoes. Only upon approval by the ICCSP may test results be accepted and applied for certification purposes.
ICCSP-approved laboratories must comply with a code of conduct approved and endorsed by the ICCSP. The code of conduct supports the integrity of laboratories, limits undue pressure, gives guidance to personnel to achieve compliance with the Scheme and requirements from the ICCSP, and promotes the role of PLS in advancing the use of seed potatoes.
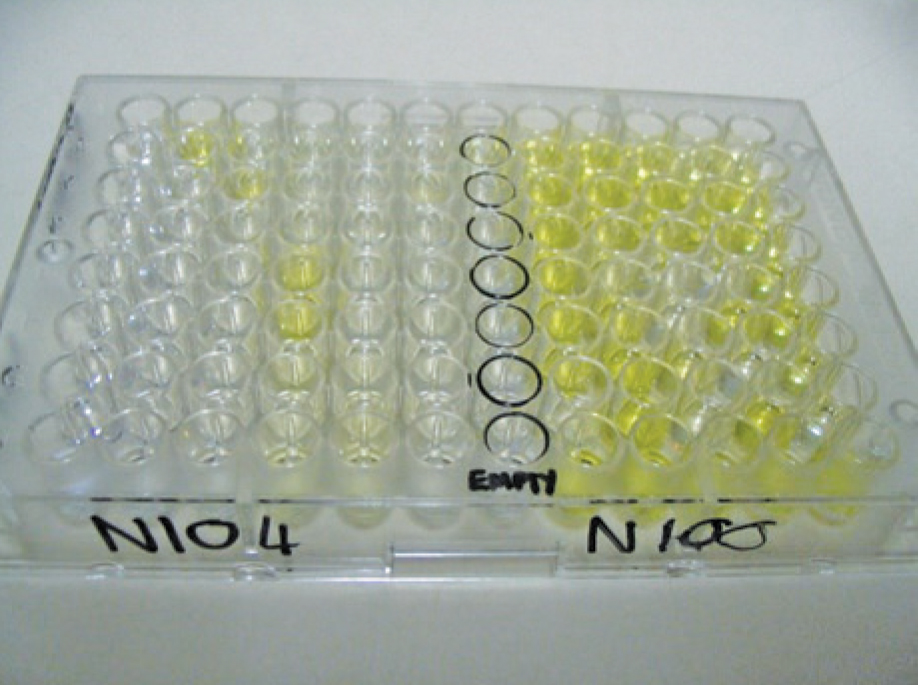
All laboratories are involved in the testing of statistically taken tuber field samples, leaf samples and diagnostic samples for the presence of PVY, PLRV and Ralstonia spp. by the execution of pathogen-specific enzyme-linked immunosorbent assay (ELISA) methods.
Field samples
Field samples are defined in the Scheme as “a representative sample of seed potatoes taken in accordance with the provisions of the protocol (of the Potato Certification Service or PCS) for the determination of the occurrence of Ralstonia spp. and viruses listed in Table 1, once the top growth on the unit concerned has died off or has been destroyed but before the seed potatoes have been removed from the soil”.
Certification officials sample separate sets of tubers (samples) from fields registered for certification. The number of tubers sampled is mostly determined by factors such as the generation of seed potatoes planted, the size of the field or unit and the status of the pathogen tested for.
However, the authority may determine otherwise. Samples for virus testing from fields planted with G0 to G2 (G1 to G3 outcome) seed tubers are 400 tubers in size, while samples taken from fields planted with G3 to G6 (G4 to G7 outcome) and certified commercial seed tubers are 200 tubers in size. Samples for testing for the presence of Ralstonia spp. consist of 4 605 tubers.
After receiving these samples at the respective laboratories (Plantovita, KwaZulu-Natal Laboratory services, Sandveld Laboratoriumdienste, GWK Aartappellaboratorium, Wesgrow Potato Laboratory), they are tested using the approved standardised methods. All tubers contained in these samples are processed and tested. The enormity of the samples makes it unique for the participating laboratories to apply all the methods employed.
Pooling of a pre-determined number of tubers in virus determinations is done for practical and financial purposes. Statistical methods are subsequently used to calculate the number of virus-infected tubers in a quantitative test process, while the regulatory status of Ralstonia spp. necessitates a qualitative test process to determine the presence or absence of the pathogen.
For field sample testing, pathogen-specific ELISA methods are applied by all laboratories. Per implication, since the inception of PLS, all laboratories have been equipped to execute the ELISA method on field samples. The robust method and specificity and sensitivity make it a simple and cost-effective method to use on a high throughput scale.
Leaf samples
Leaf samples for the determination of PVY and PLRV infection levels in planting were implemented by the authorities in 2004 as a management tool for growers, allowing a grower to sample a statistical number of leaves from a registered unit and submit the sample to a participating laboratory for testing.
The result of a leaf sample indicates the risk of virus presence in the tubers from the associated unit. However, accurately predicting the virus content in the tubers using leaf samples is not possible. Leaf samples are not considered official samples in the certification process, since the results cannot and are not used for certification purposes.
Diagnostic samples
These are samples from any kind of potato planting, taken on an ad hoc basis for testing at participating laboratories for the presence of PVY and PLRV. These samples may consist of leaves or tubers, with a maximum number of 20 leaves or tubers. If tubers are to be tested using ELISA, they must first sprout to enable accurate testing.
Diagnostic samples serve to facilitate the diagnosis of symptoms and the subsequent management of the identified disease. These samples are generally used to confirm the identification of symptoms.
Additional testing methods
In addition to the core functions of participating laboratories, Plantovita has also been equipped since the start of PLS, to test samples with conventional microbiological methods for the presence of bacterial and
fungal pathogens.
Conventional methods are employed for testing in vitro plantlets, mini tubers (generation 0, G0), and diagnostic samples for bacterial and fungal pathogens, as well as confirming the presence of Ralstonia spp. in field samples that tested positive during the execution of the ELISA. These sample types and the conventional methods require equipment different from that used in the ELISA. Interpreting the method outcomes requires additional competence that cannot be duplicated in all laboratories due to the limited number of samples.
In 2014, a validated quantitive reverse transcription polymerase chain reaction (qRT-PCR) method was implemented at Plantovita for testing field samples for the presence of PVY and PLRV. The validation work was funded by Potatoes SA and the outcome was a method established in PLS available to participating laboratories. The validation of this method was extensive due to the use thereof in the certification process on official samples, the size of samples, and the fact that it is not only used for diagnostic purposes.
The validated PVY and PLRV-specific qRT-PCR was subsequently extended for use in in vitro samples; G0 samples and diagnostic samples. qRT-PCR methods were also developed and implemented for the detection, identification and phylotyping of Ralstonia spp. and corky ringspot (CRS) causal viruses (tomato spotted wilt, potato mop-top virus, tobacco rattle virus and pepper ringspot virus [PepRSV]).
Plantovita funded the development of the Ralstonia spp. assays, while PSA contributed funding to the CRS project. Further development of PCR-based methods is ongoing to the benefit of the potato industry and its growers. – Anel Espach, technical manager, Plantovita
For more information, contact Plantovita at info@plantovita.co.za or phone 012 819 8100.
